 Antibody and Protein Expression in CHO and Cell Line Development
Antibody and Protein Expression in CHO and Cell Line Development Antibody or Fc-fusion Protein Expression in CHO
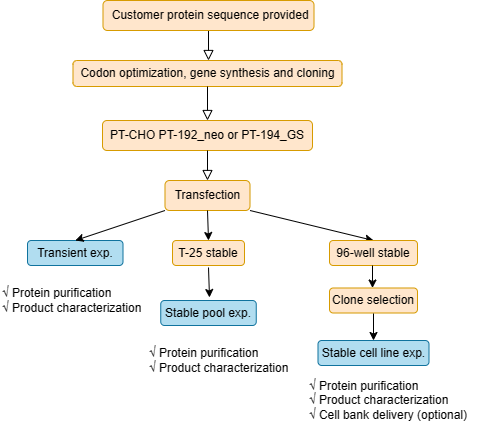
CHO cells account for >70% of recombinant biopharmaceutical protein productions due to their ability to express complex proteins with proper folding and posttranslational modification (PTM). Our proprietary PT-CHO mammalian expression system includes the PT-CHO host cells and highly efficient mammalian expression vectors (PT-192_neo and PT-194_GS). The cells can grow prolifically in suspension with serum-free and chemically defined medium, allowing volumetric scalability and resulting high cell density and high titer. The PT-CHO expression system facilitates transient expression, stable pool expression, and the development of stable cell lines for stable protein expression in order to meet our clients’ needs.
The service begins with an antibody or a protein sequence provided by the client. We carry out gene synthesis with codon optimization, expression and purification, scale up and product characterization, and deliver to the client the expressed, purified, and characterized antibody or protein products. In addition, we can optionally deliver the expression cell lines. Furthermore, if requested, we are capable of providing the expression cell lines.
Please contact us for your protein expression inquiries or requests.
Application of protein expression service:
1. The expression of antibodies or other proteins of interest (such as Fc-fusion proteins or drug target proteins) with known gene or protein sequences.2. The expression of a recombinant antibody by determining its sequence using our Antibody De Novo Sequencing service, such as that of a commercially available antibody. We only require 200ug-400ug antibody sample to obtain 100% antibody sequences with almost 100% accuracy. For the expression of a recombinant antibody, the Fc region of the antibody can be changed to suitable species and suitable types. We can also provide the services to humanize the variable regions as well as offering affinity maturation based on the de novo sequencing results.
3. This platform is an essential part of our Therapeutic Antibody Discovery and Development service, in which a drug target protein is expressed and used to immunize animals to generate hybridomas and specific neutralizing antibodies. After, a number of recombinant antibodies with different amino acids sequences (possibly different epitopes) are produced in functional studies.
4. This platform is an essential part of our Neutralizing Antibody Drug Discovery and Development services. Typically, 10-20 high affinity antibodies are purified from the infected patient serum samples, de novo sequenced, and then expressed in CHO cells to test for neutralizing activity.
Related Service & Products